How do you measure TOC?
Simply put, the job of a TOC analyzer is to oxidize organic compounds into CO2 and measure the amount of CO2 produced. Regardless of the oxidation technique, TOC must be determined by either measuring or removing the various fractions found within the sample's total carbon (TC). These fractions are defined below:
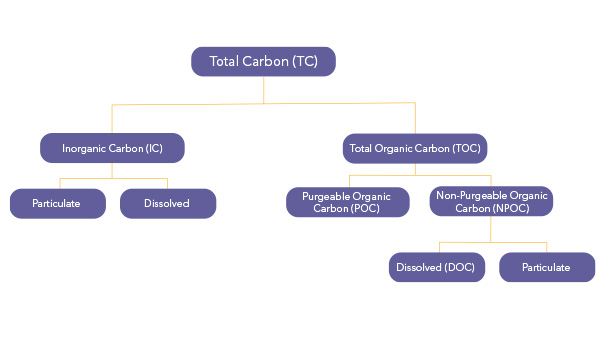
Total Organic Carbon (TOC) — all carbon atoms covalently bonded in organic molecules
Inorganic Carbon (IC) — carbonate, bicarbonate, and dissolved CO2
Dissolved Organic Carbon (DOC) — the fraction of TOC that passes through a 0.45 μm-pore-diameter filter
Suspended Organic Carbon (also called Particulate Organic Carbon) — the fraction of TOC retained by a 0.45- μm filter
Purgeable Organic Carbon (POC, sometimes called Volatile Organic Carbon) — the fraction of TOC removed from an aqueous solution via gas stripping under specified conditions
Nonpurgeable Organic Carbon (NPOC) — the fraction of TOC not removed by gas stripping. In most cases, inorganic carbon is purged and not "determined," in which case only NPOC is determined, and POC is assumed to be negligible.
This graphic offers a visual of how these fractions of TC are divided:
Other Important Terms:
Reagent Blank – The detector response is generated from an analysis sequence with reagents without introducing a sample or standard. This response is due to carbon contamination in the reagents, gas, digestion vessel, and/or tubing.
Standard – Any sample with a known amount of added carbon
Water Blank or Instrument Method Blank – Response of an analyzer to the carbon content of the water. This is measured by using water as the zero-concentration standard during calibration.